Lots of interesting abstracts and cases were submitted for TCTAP 2026. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge!
CASE20251114_048
Transcatheter Solutions for the Holes
By Krissada Meemook, Keratisorn Pakpian, Mann Chandavimol
Presenter
Keratisorn Pakpian
Authors
Krissada Meemook1, Keratisorn Pakpian2, Mann Chandavimol1
Affiliation
Ramathibodi, Thailand1, Ramrthibodi, Thailand2
View Study Report
CASE20251114_048
Structural - Other Structural Interventions
Transcatheter Solutions for the Holes
Krissada Meemook1, Keratisorn Pakpian2, Mann Chandavimol1
Ramathibodi, Thailand1, Ramrthibodi, Thailand2
Clinical Information
Relevant Clinical History and Physical Exam
A 76-year-old man with hypertension, dyslipidemia, chronic kidney disease stage 3, and atrial fibrillation was a known case of severe mitral regurgitation due to anterior mitral valve prolapse with a flail leaflet. He underwent mitral valve replacement (MVR) with a 25-mm bioprosthetic valve in 2023. Postoperative echocardiography demonstrated a well-seated prosthetic mitral valve (PrMV) without evidence of obstruction or regurgitation.In October 2024, he developed symptoms of clinical heart failure. Transthoracic echocardiography (TTE) showed borderline left ventricular ejection fraction (LVEF) of 50%, a well-seated bioprosthetic mitral valve, and moderate paravalvular mitral regurgitation (MR). Transesophageal echocardiography (TEE) confirmed a well-seated bioprosthetic valve without valvular obstruction and identified a paravalvular leak at the 2 o’clock position (surgical view). The defect measured 3.5 × 4.1 mm and resulted in moderate MR with an effective regurgitant orifice area (EROA) of 0.35 cm² and regurgitant volume of 56 mL, along with trace intravalvular regurgitation.Despite optimized medical therapy, the patient remained symptomatic. A heart team discussion concluded that redo surgery was high risk due to multiple comorbidities, with an STS-predicted mortality >10%. Therefore, percutaneous paravalvular leak (PVL) closure was recommended.In March 2025, the patient presented with severe headache, projectile vomiting, and nocturnal pain. CT brain revealed bilateral subdural hematomas (SDH), and laboratory testing showed an INR of 8.31. He underwent right burr hole and craniotomy with clot evacuation. He was subsequently left with two major ongoing clinical issues: moderate paravalvular mitral regurgitation with persistent heart failure, and paroxysmal atrial fibrillation with a history of life-threatening intracranial bleeding, making long-term anticoagulation unsafe.A combined transcatheter intervention was successfully performed. First, transcatheter closure of the mitral PVL was achieved using a single Amplatzer Vascular Plug III (AVP III) device. This was followed by left atrial appendage (LAA) occlusion using a Lambre device sized 38/24 mm to reduce thromboembolic risk without the need for extended anticoagulation.Follow-up echocardiography demonstrated a well-seated PVL closure device with only trace residual paravalvular regurgitation. The LAA occluder was well positioned, with a small posterior peridevice leak measuring up to 1.2 mm. The mitral bioprosthesis remained well seated with mild intravalvular regurgitation.This case highlights the feasibility and clinical benefit of a combined transcatheter approach for managing complex post-MVR complications and stroke prevention in a high-surgical-risk patient.
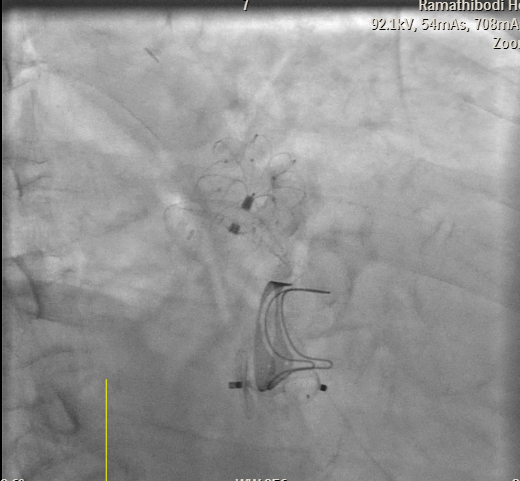
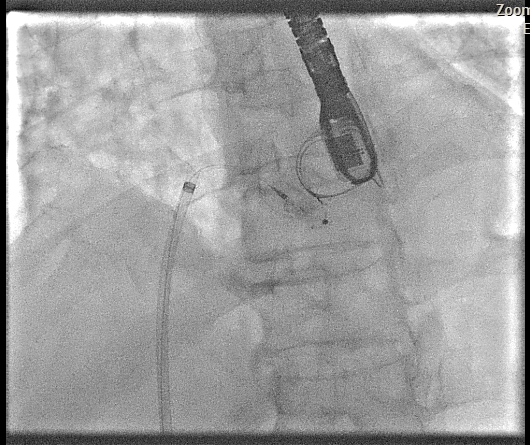
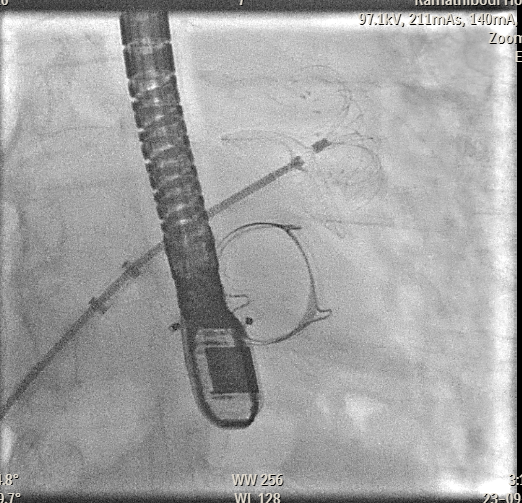
 2 (7).mp4
2 (7).mp4
 3 (7).mp4
3 (7).mp4
 4 (7).mp4
4 (7).mp4



Relevant Test Results Prior to Catheterization
Relevant Catheterization Findings
Interventional Management
Procedural Step
Transesophageal Echocardiography (TEE) EvaluationTEE was performed and demonstrated severe paravalvular leakage of the bioprosthetic mitral valve at the 3 o’clock position.Vascular Access PreparationBoth groins were infiltrated with 10 mL of 2% lidocaine for local anesthesia. Using the front-wall technique, a 6F sheath was inserted into the right femoral vein and subsequently upsized to a 12F FastCath introducer sheath. The left femoral artery was cannulated with a 6F sheath for continuous arterial pressure monitoring.Transseptal PunctureA 0.032-inch guidewire was advanced into the superior vena cava, followed by placement of an 8.5F SL1 introducer. A BRK 18-gauge transseptal needle was introduced with the needle tip kept within the sheath.Under TEE and fluoroscopic guidance, the system was slowly withdrawn into the right atrium to identify the optimal puncture site. A successful mid-posterior transseptal puncture was achieved into the left atrium.Hemodynamics recorded LA pressure of 12 mmHg with a peak V-wave of 19 mmHg.Crossing the Paravalvular Leak (PVL)A 0.032-inch wire was advanced across the PVL, and the 8.5F Agilis catheter was positioned in the left atrium.Wire and Catheter Exchange for PVL CrossingA 6F MP01 guiding catheter was directed toward the medial aspect of the mitral valve. A 0.035-inch Terumo wire crossed into the left ventricle and was exchanged for an Amplatz Super Stiff wire.Sheath ExchangeThe 6F MP guiding catheter and 8.5F Agilis sheath were removed and replaced with a 7F DuraSheath (90 cm) to provide adequate support.First Attempt at PVL ClosureAn Amplatzer Vascular Plug III (AVP III) 5 × 10 mm was delivered across the defect. Despite optimal positioning, the device did not achieve complete occlusion of the PVL.Successful PVL ClosureThe Trevisio 8F delivery system replaced the 7F DuraSheath. A larger AVP III 5 × 14 mm device was advanced across the defect and deployed.After position adjustment, the device was released. TEE confirmed a stable device position with no residual PVL. All equipment was removed, and the venous access was upsized to a 14F FastCath introducer to prepare for LAA occlusion.Transition to LAA Occlusion PhaseA 10F Lambre delivery sheath was advanced into the left upper pulmonary vein (LUPV), replacing the SL1 sheath.Accessing the LAAA 5F pigtail catheter was inserted, and the delivery sheath was advanced into the LAA under fluoroscopic and TEE guidance.Device PreparationA 38/34 mm Lambre LAA occlusion device was prepared and loaded into the delivery system.LAA Occlusion DeploymentThe device was deployed just proximal to the LAA ostium. Stability and seating were confirmed using angiography and TEE.LAA angiography demonstrated trace residual flow into the LAA. The device was then detached.Real-time 3D TEE confirmed proper positioning with no significant peridevice leak.Hemostasis and CompletionAll delivery systems were removed. Venous hemostasis was achieved using a figure-of-eight suture, and arterial access was sealed with manual compression. The procedure was completed without immediate complications.
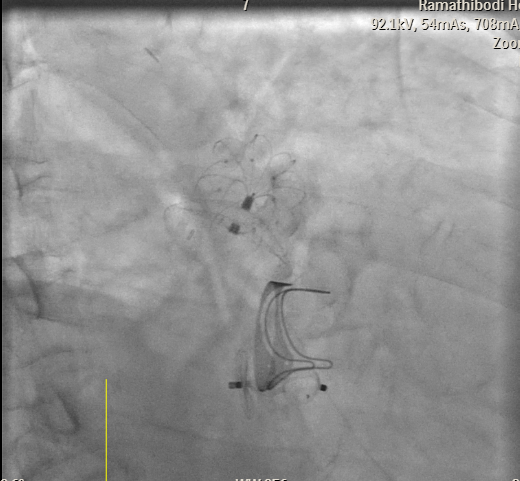
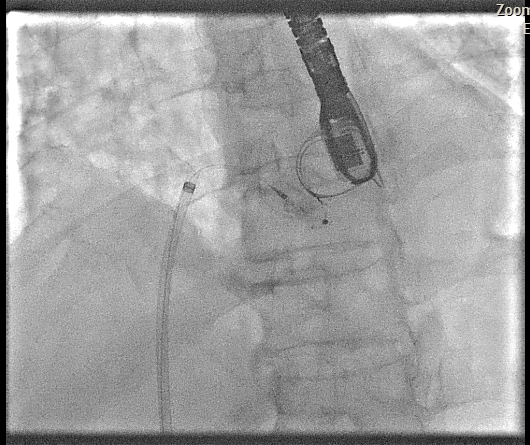
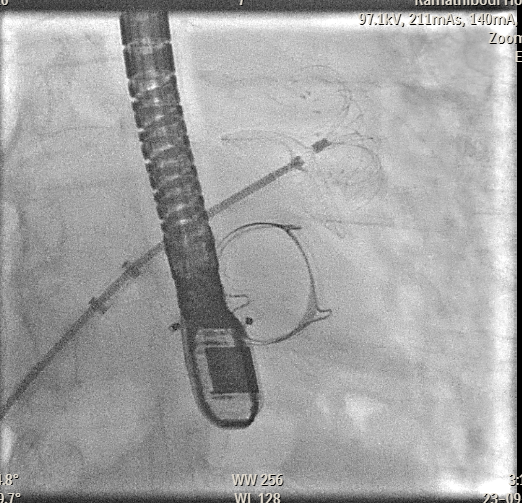



Case Summary
This case illustrates the effectiveness of a combined transcatheter approach for managing dual structural complications—paravalvular mitral regurgitation and left atrial appendage–related thromboembolic risk—in a high-surgical-risk patient after bioprosthetic mitral valve replacement. The successful use of an Amplatzer Vascular Plug III for PVL closure and a Lambre device for LAA occlusion resulted in significant symptomatic improvement, minimal residual leaks, and avoidance of reoperation or long-term anticoagulation. This experience emphasizes the role of percutaneous interventions as a safe and feasible alternative to redo surgery in elderly patients with multiple comorbidities and elevated bleeding risk.
