Lots of interesting abstracts and cases were submitted for TCTAP 2026. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge!
ABS20251111_0003
Limus-Coated vs Paclitaxel-Coated Balloon for Percutaneous Coronary Intervention: Updated Systematic Review and Meta-Analysis of Randomized Controlled Trials
By Pirel Aulia Baravia, Bernadus Bernardino Bramantyo, Ganiera Putri Barabanuas Nurfairudz, Dicky Armein Hanafy, Isman Firdaus
Presenter
Pirel Aulia Baravia
Authors
Pirel Aulia Baravia1, Bernadus Bernardino Bramantyo2, Ganiera Putri Barabanuas Nurfairudz3, Dicky Armein Hanafy1, Isman Firdaus1
Affiliation
National Cardiovascular Center Harapan Kita, Jakarta, Indonesia1, Sardjito General Hospital, Indonesia2, Universitas Brawijaya, Indonesia3
View Study Report
ABS20251111_0003
DES/BRS/DCB
Limus-Coated vs Paclitaxel-Coated Balloon for Percutaneous Coronary Intervention: Updated Systematic Review and Meta-Analysis of Randomized Controlled Trials
Pirel Aulia Baravia1, Bernadus Bernardino Bramantyo2, Ganiera Putri Barabanuas Nurfairudz3, Dicky Armein Hanafy1, Isman Firdaus1
National Cardiovascular Center Harapan Kita, Jakarta, Indonesia1, Sardjito General Hospital, Indonesia2, Universitas Brawijaya, Indonesia3
Background
Although the meta-analysis of limus-based coated balloon (LCB) and paclitaxel coated balloon (PCB) have previously investigated, a reevaluation of safety and efficacy is needed with recent randomised controlled trials (RCT)
Methods
Systematic database searching for randomized controlled trials (RCTs) up to October 2025. The primary outcomes were target lesion revascularization (TLR) while secondary outcomes are clinical and angiographic outcomes. Pooled relative risk (RR) and mean difference (MD) with 95% confidence intervals (CI) were calculated using random-effects models.

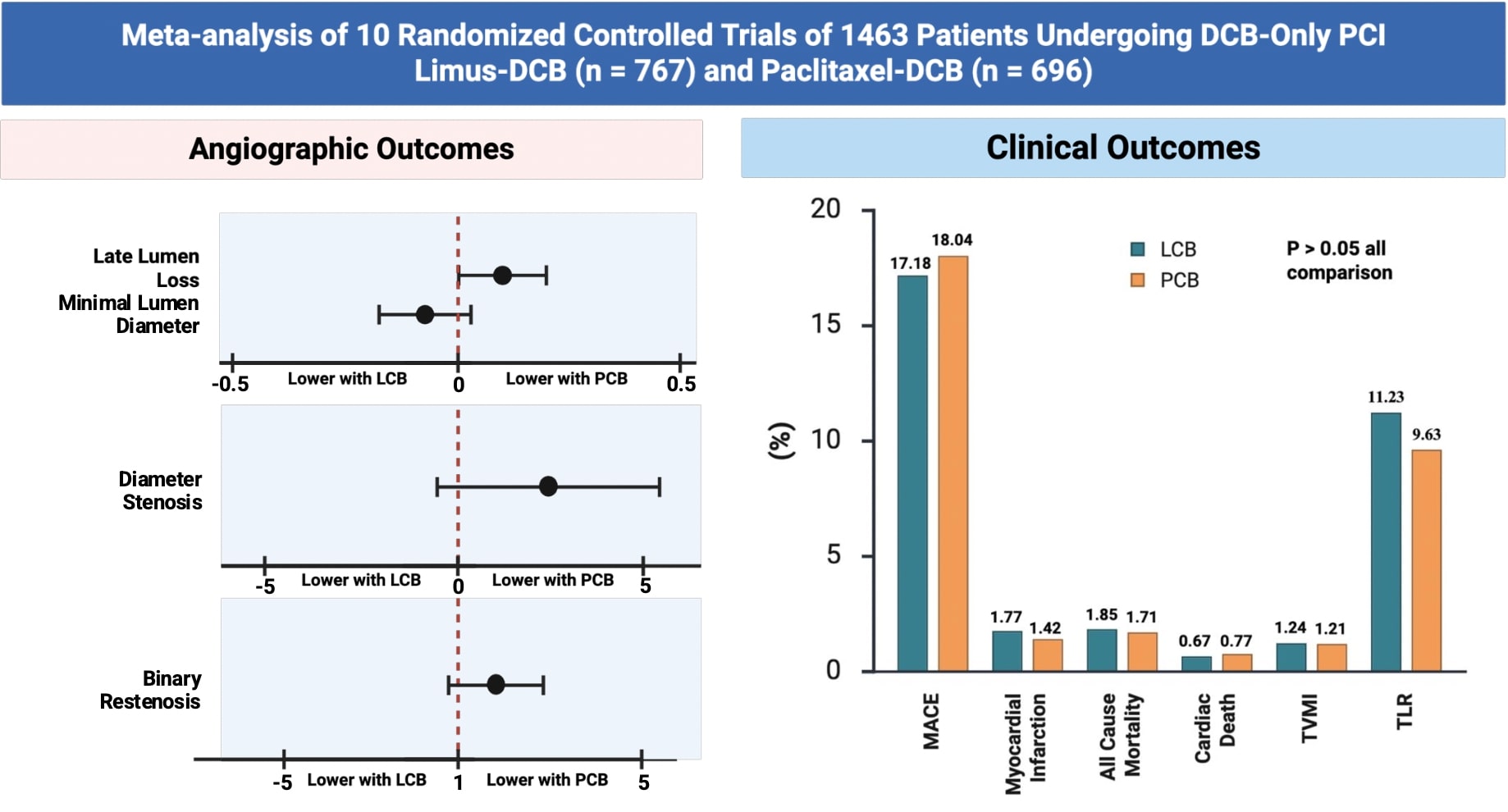
Results
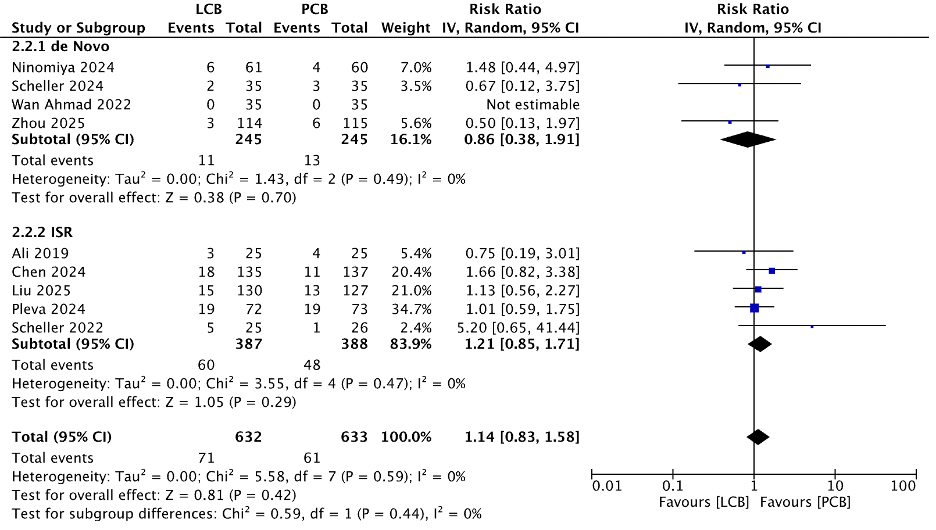
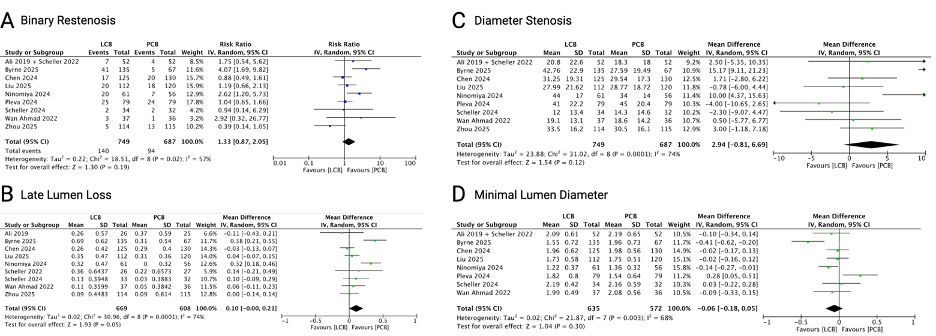
Ten RCTs with 1463 patients and 1434 lesions were included in the analysis. LCB was comparable with PCB in TLR (RR 1.14; 0.83 - 1.58; p = 0.42), MACE (RR 0.98; 95% CI 0.64 - 1.48; p = 0.91), myocardial infarction (RR 1.22; 95% CI 0.47 - 3.17; p = 0.69), all-cause mortality (RR 1.04; 95% CI 0.40 - 2.70; p = 0.94), cardiac death (RR 0.73; 95% CI 0.17 - 3.22; p = 0.68), and TVMI (RR 0.71; 95% CI 0.28 - 1.84; p = 0.49). Angiographic follow-up showed comparable results between LCB and PCB evaluated from binary restenosis (RR 1.33; 95% CI 0.87 - 2.05; p = 0.19), LLL (MD 0.10; 95% CI -0.00 to 0.21; p = 0.05), diameter stenosis (MD 2.94; 95% CI -0.81 to 6.69; p = 0.12), MLD (MD -0.09; 95% CI -0.18 to 0.05; p = 0.30).
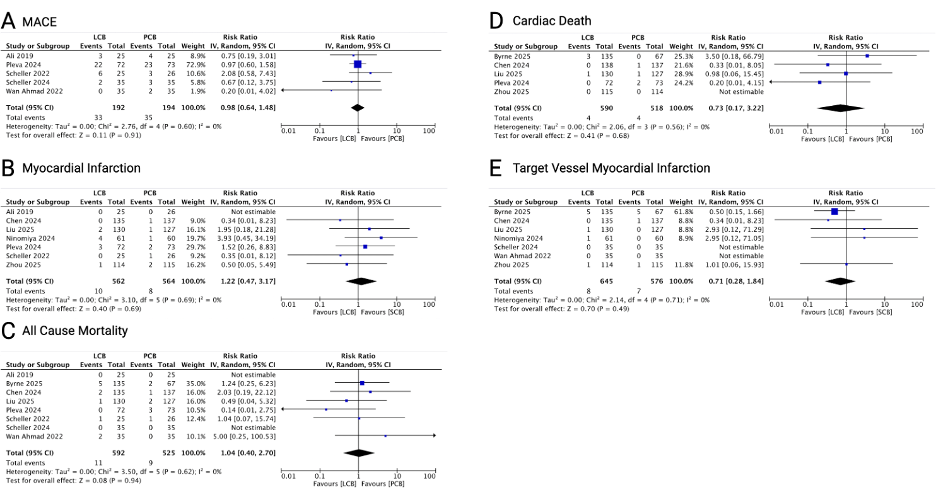



Conclusion
Limus-coated balloon demonstrated a comparable safety profile with Paclitaxel-coated balloon according to clinical outcomes and angiographic assessments.