Lots of interesting abstracts and cases were submitted for TCTAP 2026. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge!
ABS20251101_0003
Impact of Peri-TAVI GLP-1 Receptor Agonist Therapy on Mortality and Heart Failure: Evidence From a Global Real-World Database
By Yu Chuan Chuang, Ming-Ju Chuang, Chih-Yen Wang, Tsun-Jui Liu, Wen-Lieng Lee
Presenter
Yu Chuan Chuang
Authors
Yu Chuan Chuang1, Ming-Ju Chuang1, Chih-Yen Wang1, Tsun-Jui Liu1, Wen-Lieng Lee1
Affiliation
Taichung Veterans General Hospital, Taiwan1
View Study Report
ABS20251101_0003
Vascular Heart Disease and Cardiomyopathies
Impact of Peri-TAVI GLP-1 Receptor Agonist Therapy on Mortality and Heart Failure: Evidence From a Global Real-World Database
Yu Chuan Chuang1, Ming-Ju Chuang1, Chih-Yen Wang1, Tsun-Jui Liu1, Wen-Lieng Lee1
Taichung Veterans General Hospital, Taiwan1
Background
Transcatheter aortic valve implantation (TAVI) has become an established therapy for patients with severe aortic stenosis (AS) across the spectrum of surgical risk. Despite significant advances, post-TAVI mortality and morbidity remain substantial.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), such as semaglutide and liraglutide, have demonstrated cardiovascular benefits in patients with diabetes and high cardiovascular risk.
However, their impact on outcomes following TAVI has not been systematically studied, as this population has often been excluded from major clinical trials.
We therefore sought to evaluate the association between GLP-1 RA use and clinical outcomes in patients undergoing TAVI using a large, real-world dataset.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), such as semaglutide and liraglutide, have demonstrated cardiovascular benefits in patients with diabetes and high cardiovascular risk.
However, their impact on outcomes following TAVI has not been systematically studied, as this population has often been excluded from major clinical trials.
We therefore sought to evaluate the association between GLP-1 RA use and clinical outcomes in patients undergoing TAVI using a large, real-world dataset.
Methods
We used the TriNetX research network to identify adults (≥18 years) with rheumatic or non-rheumatic severe aortic stenosis who underwent transcatheter aortic valve implantation (TAVI) between January 2010 and January 2024. Patients were excluded if they had pure aortic regurgitation, prior surgical aortic valve replacement, bariatric surgery within 6 months, end-stage renal disease, or malignant neoplasm. Patients were divided into two groups: (1) the GLP-1 receptor agonist (GLP-1 RA) group, defined as those prescribed semaglutide, liraglutide, dulaglutide, exenatide, or lixisenatide within 1 month before or up to 2 weeks after TAVI; and (2) the control group, defined as those who never received a GLP-1 RA from baseline through 1 year after TAVI. To reduce confounding, 1:1 nearest-neighbor propensity score matching without replacement was performed. Matching variables included demographics (age, sex, race), comorbidities (type 1 or 2 diabetes, hypertension, heart failure, ischemic heart disease, hyperlipidemia, atrial fibrillation, chronic kidney disease, cerebrovascular disease, peripheral vascular disease, COPD), medications (antiplatelets, anticoagulants, antihypertensives, diuretics, statins, glucose-lowering therapies, corticosteroids), and laboratory parameters (creatinine, eGFR, HbA1c, BMI, natriuretic peptides, LVEF). Standardized mean differences (SMD) <0.1 were considered well balanced. Follow-up began 14 days after TAVI and continued to 365 days. The primary outcome was all-cause mortality. Secondary outcomes were worsening heart failure, all-cause hospitalization, major adverse cardiovascular events, acute kidney injury, and pacemaker implantation. Safety outcomes were pancreatitis and diabetic ketoacidosis; nephrolithiasis served as a falsification endpoint. Kaplan–Meier curves with log-rank tests evaluated event-free survival. Cox proportional hazards models estimated hazard ratios with 95% confidence intervals. A two-sided p<0.05 was considered statistically significant. Analyses were performed using the TriNetX Analytics platform with de-identified electronic health record data.
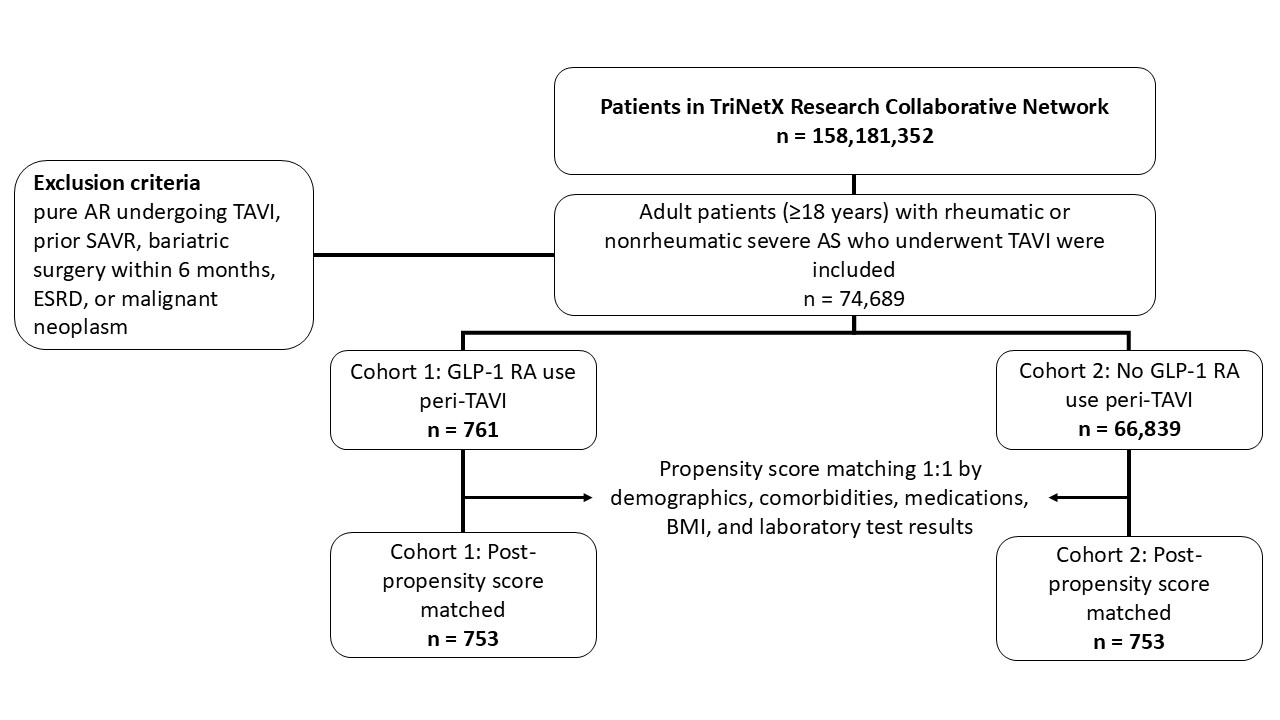

Results
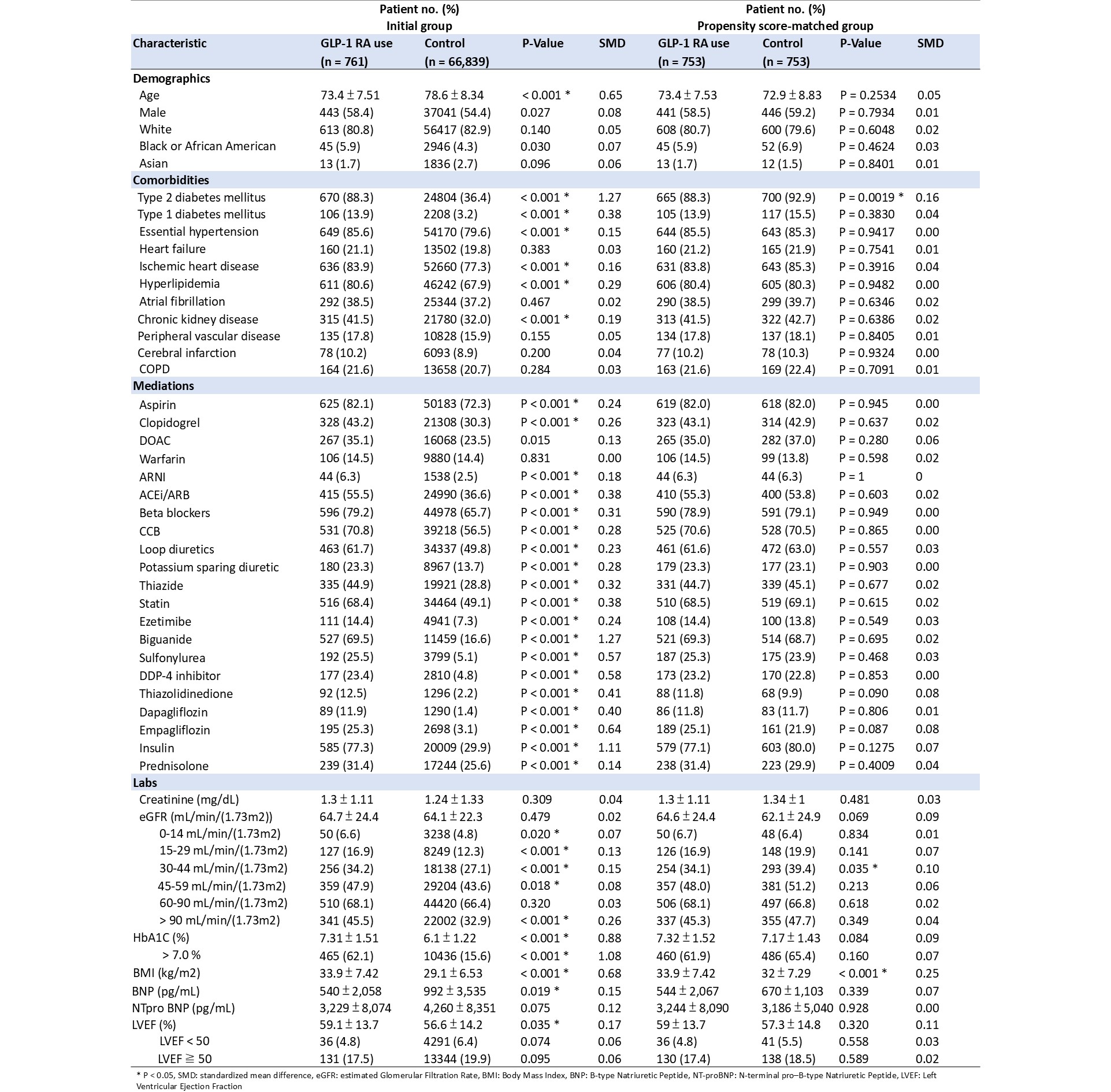
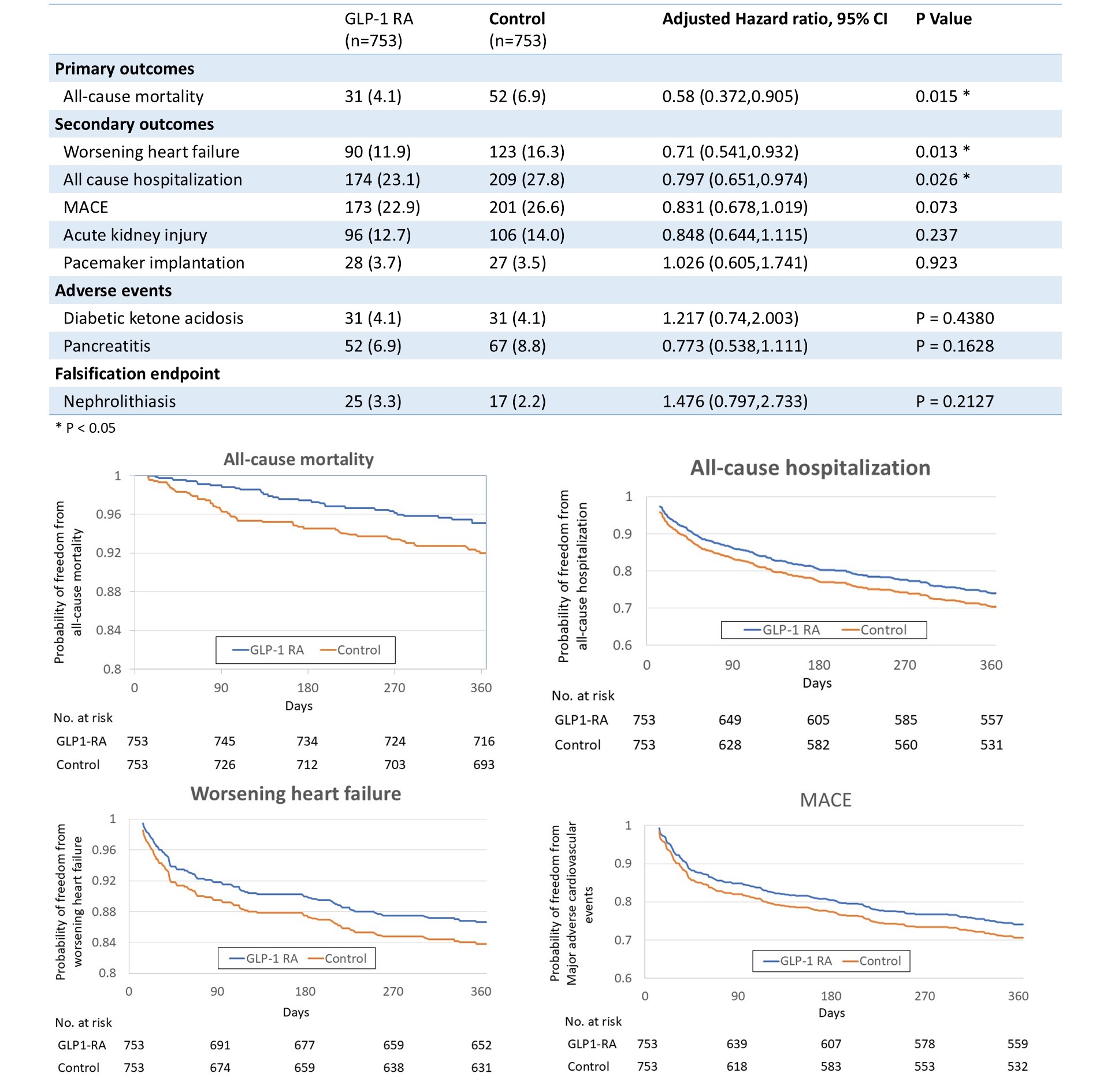
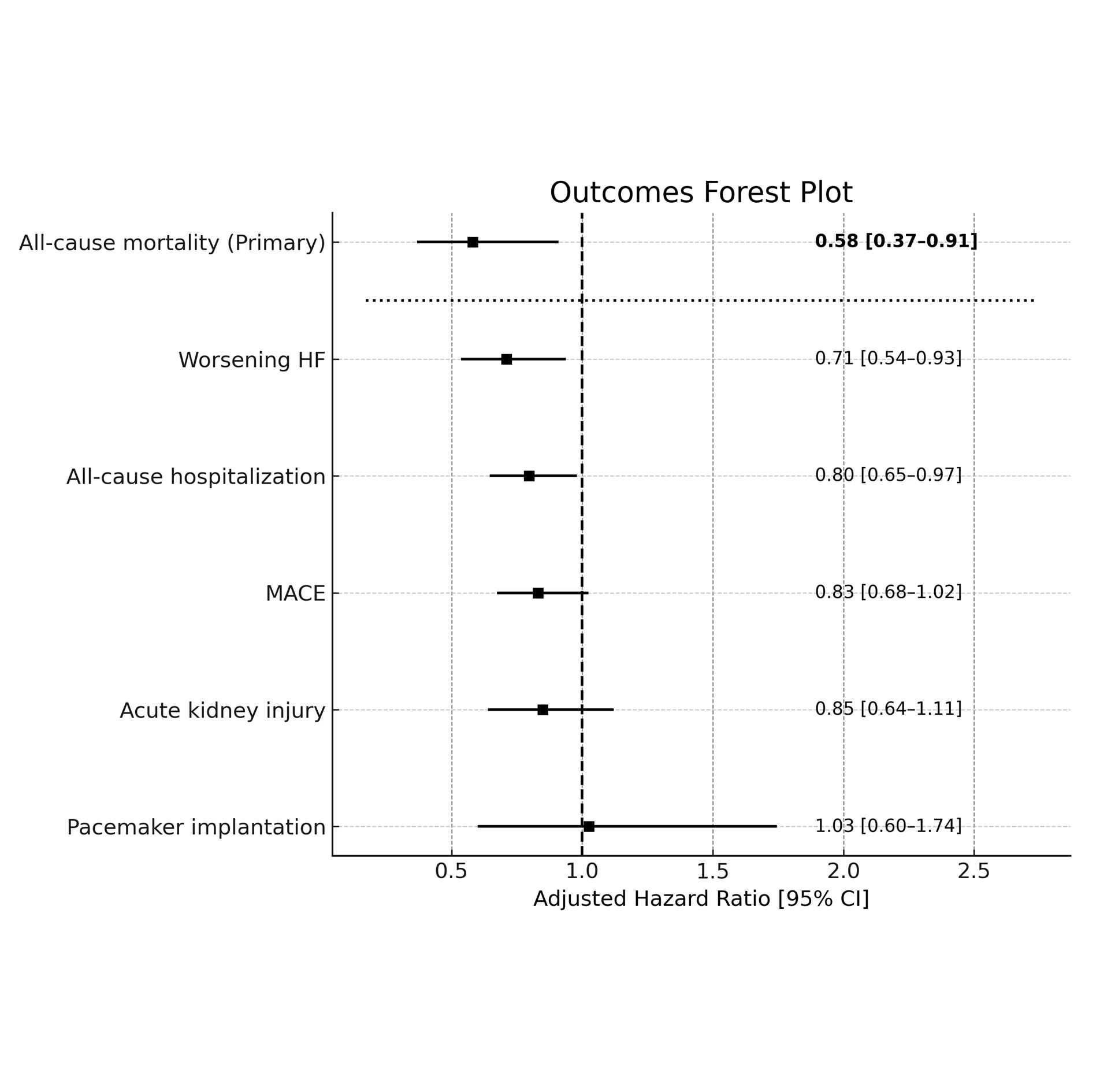
A total of 761 patients received a GLP-1 RA and 66,839 served as controls. After 1:1 propensity score matching, baseline demographics and most comorbidities were well balanced between groups. Residual differences included a higher prevalence of type 2 diabetes among GLP-1 RA users (92.9% vs 88.3%, p=0.0019), a greater proportion of patients with moderate CKD (eGFR 30–44 mL/min/1.73m²) in the control group (39.4% vs 34.1%, p=0.0359), and higher BMI in the GLP-1 RA cohort (33.9 ± 7.42 vs 32.0 ± 7.29 kg/m², p<0.0001). During follow-up from day 14 to 1 year after TAVI, the primary endpoint of all-cause mortality was significantly lower in the GLP-1 RA group (4.1% vs 6.9%; adjusted HR 0.58, 95% CI 0.372–0.905; p=0.0151). GLP-1 RA use was also associated with reduced worsening heart failure (11.9% vs 16.3%; aHR 0.71, 95% CI 0.541–0.932; p=0.0131) and lower all-cause hospitalization (23.1% vs 27.8%; aHR 0.797, 95% CI 0.651–0.974; p=0.0263). There was a nonsignificant trend toward lower MACE (22.9% vs 26.6%; aHR 0.831, 95% CI 0.678–1.019; p=0.0739). Rates of acute kidney injury (12.7% vs 14.0%; aHR 0.848, p=0.2371) and pacemaker implantation (3.7% vs 3.5%; aHR 1.026, p=0.9235) did not differ between groups. Adverse events, including pancreatitis and diabetic ketoacidosis, were rare and comparable. The falsification endpoint of nephrolithiasis showed no difference, supporting the robustness of the findings.



Conclusion
In this large real-world analysis, peri-procedural GLP-1 RA use in patients undergoing TAVI was associated with improved clinical outcomes, including lower all-cause mortality, reduced risk of worsening heart failure, and fewer hospitalizations at 1 year. The observed mortality benefit (aHR 0.58) is consistent with prior cardiovascular outcome trials of GLP-1 RAs in diabetic populations, but our study extends these findings to the structural heart disease setting. The reduction in heart failure events may reflect the pleiotropic metabolic and cardioprotective effects of GLP-1 RAs, including weight loss, improved glycemic control, reduced inflammation, and potential direct myocardial benefits. The absence of a significant reduction in MACE likely reflects the multifactorial pathophysiology of ischemic complications in the post-TAVI population, where procedural factors and structural anatomy may outweigh metabolic modulation. Similarly, the lack of association with acute kidney injury or conduction abnormalities suggests that GLP-1 RA–mediated protection may not influence these peri-procedural pathways. Despite comprehensive propensity matching, residual confounding remains possible, particularly given differences in diabetes prevalence, BMI, and CKD burden. The observational nature of the study precludes causal inference, and these results should be interpreted as hypothesis-generating. Overall, GLP-1 RA use around the time of TAVI was associated with lower 1-year mortality, fewer heart failure events, and reduced hospitalization without an increase in adverse events. These findings highlight a potential therapeutic role for GLP-1 RAs in optimizing post-TAVI outcomes and support the need for prospective studies or randomized trials to confirm these associations.
