Lots of interesting abstracts and cases were submitted for TCTAP 2026. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge!
ABS20251023_0002
Evaluating the Efficacy of Intravascular Ultrasound-Guided Percutaneous Coronary Intervention With Automated Longitudinal Analysis in Optimizing Coronary Stent Outcomes
By Kittipong Youpaniad, Anuruck Jeamanukoolkit, Wasant Soonfuang, Sukhum Tachasakunjareon, Sakolwat Montrivade
Presenter
Kittipong Youpaniad
Authors
Kittipong Youpaniad1, Anuruck Jeamanukoolkit2, Wasant Soonfuang2, Sukhum Tachasakunjareon2, Sakolwat Montrivade2
Affiliation
Nakhonpathom Hospital, Thailand1, Police General Hospital, Thailand2
View Study Report
ABS20251023_0002
Invasive Imaging (IVUS, OCT, NIRS, VH, etc)
Evaluating the Efficacy of Intravascular Ultrasound-Guided Percutaneous Coronary Intervention With Automated Longitudinal Analysis in Optimizing Coronary Stent Outcomes
Kittipong Youpaniad1, Anuruck Jeamanukoolkit2, Wasant Soonfuang2, Sukhum Tachasakunjareon2, Sakolwat Montrivade2
Nakhonpathom Hospital, Thailand1, Police General Hospital, Thailand2
Background
Coronary artery disease (CAD) is one of the leading causes of morbidity and mortality worldwide. Percutaneous coronary intervention (PCI) with stent implantation is a cornerstone in the treatment of CAD. However, post-procedural complications such as in-stent restenosis (ISR) and stent thrombosis continue to pose major clinical challenges. One of the key contributing factors to these complications is suboptimal stent deployment. Intravascular ultrasound (IVUS) has been developed as an imaging modality to enhance the efficacy of PCI. Evidence from previous trials such as the IVUS-XPL and ULTIMATE demonstrated that IVUS-guided PCI significantly reduced the incidence of target vessel failure (TVF). These studies established a set of Optimal IVUS Criteria for stent deployment. In the ULTIMATE trial, only 53% of patients underwent IVUS-guided PCI were able to meet all three optimal IVUS criteria, highlighting the challenges of achieving stent optimization in clinical practice. Automated longitudinal analysis technology has been developed to enhance the utility of IVUS. This system (available in commercial platforms from Boston Scientific and Insight Lifetech, etc.) automatically detects inappropriate stent or lumen area and excessive plaque burden, thereby reducing dependence on operator expertise and improving the precision of IVUS-guided PCI. The objective of this study is to evaluate stent expansion following IVUS-guided PCI with automated longitudinal analysis compared to conventional IVUS-guided PCI.
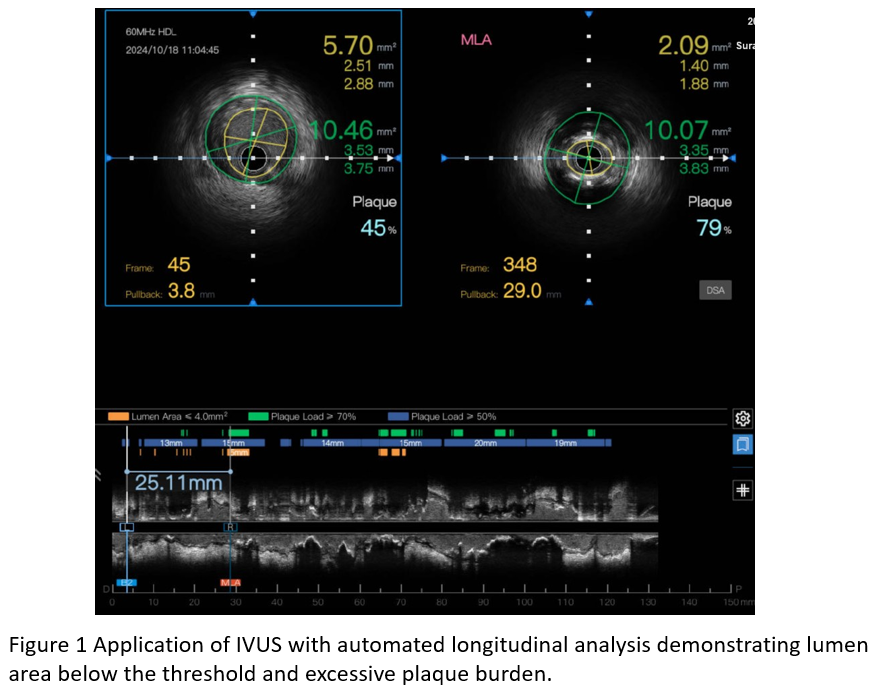
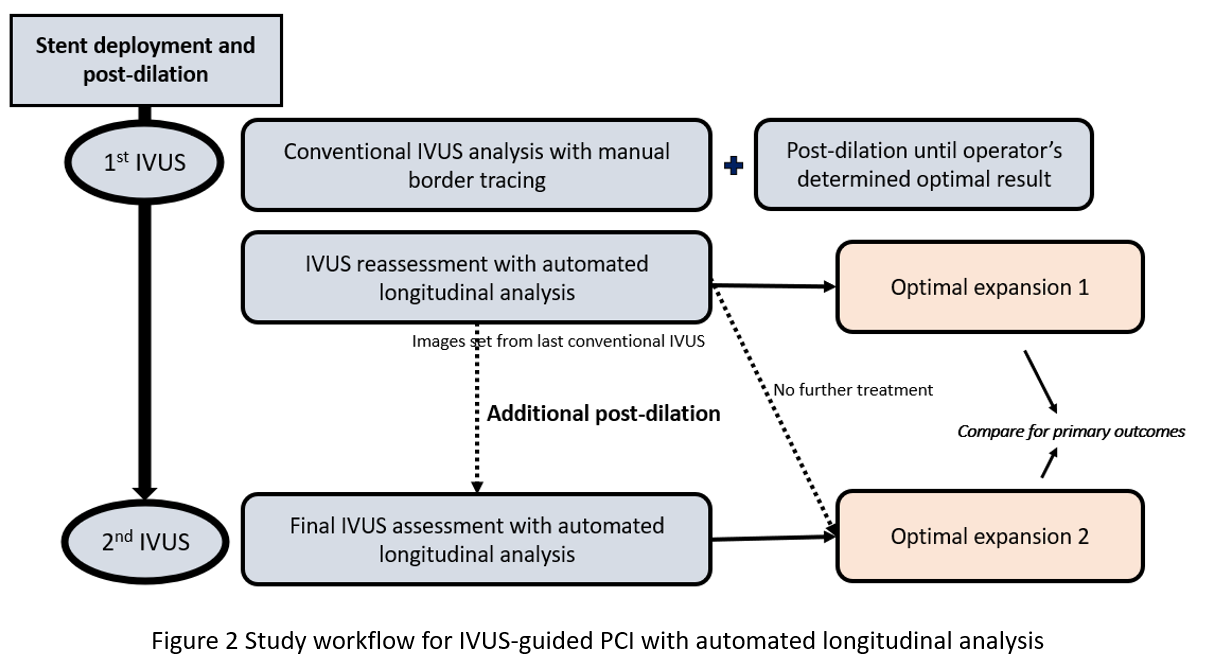



Methods
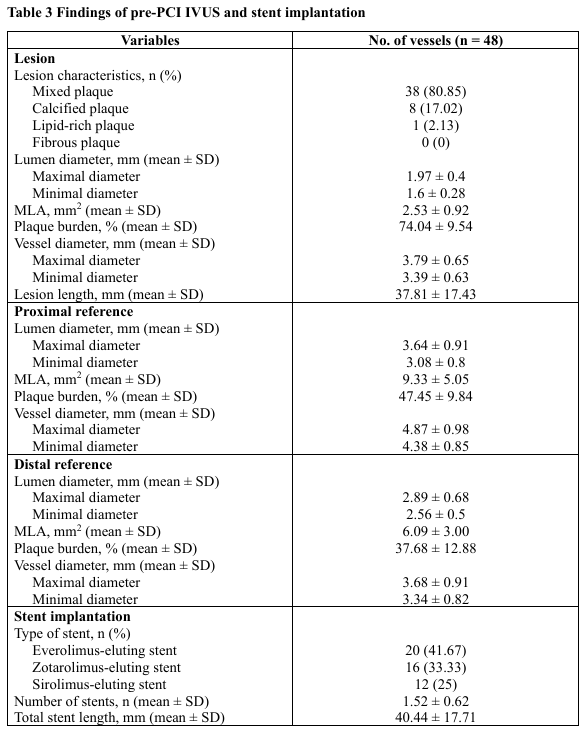
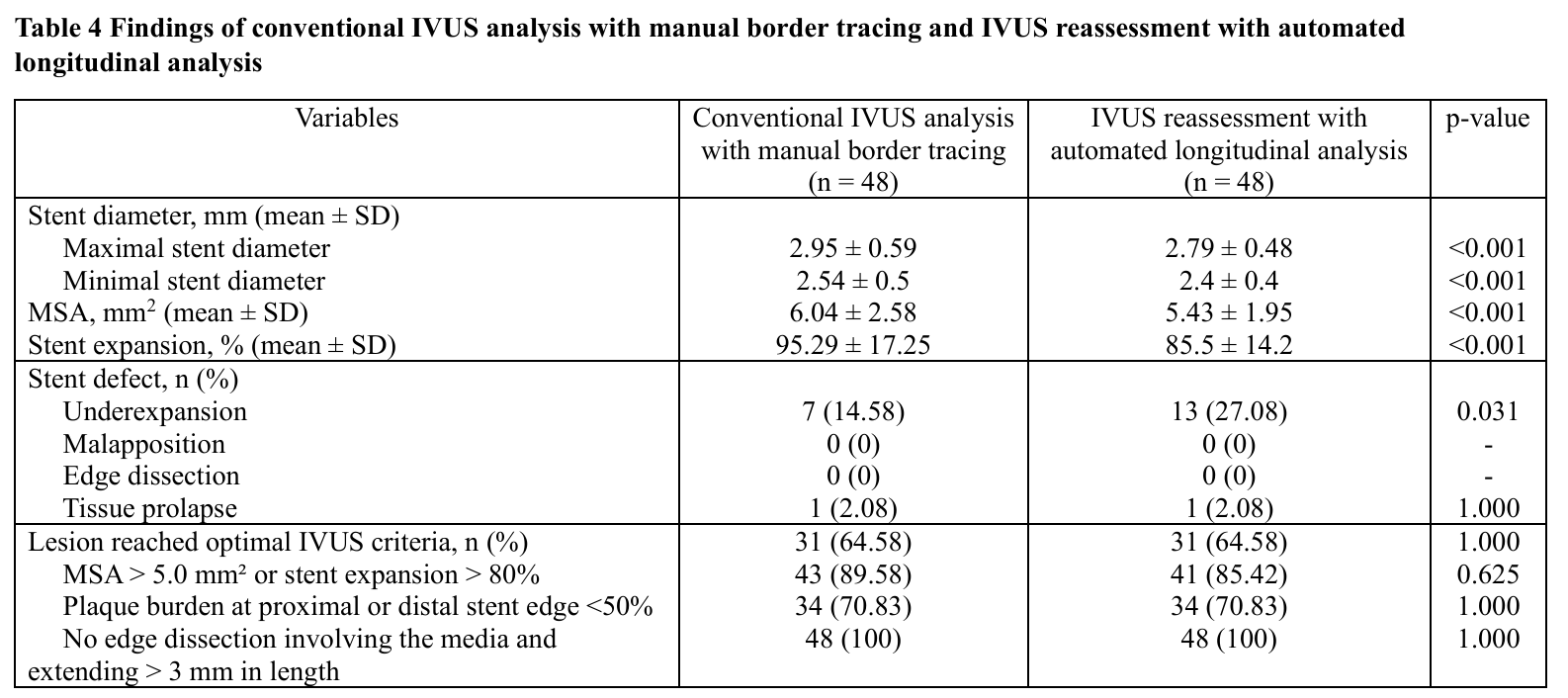
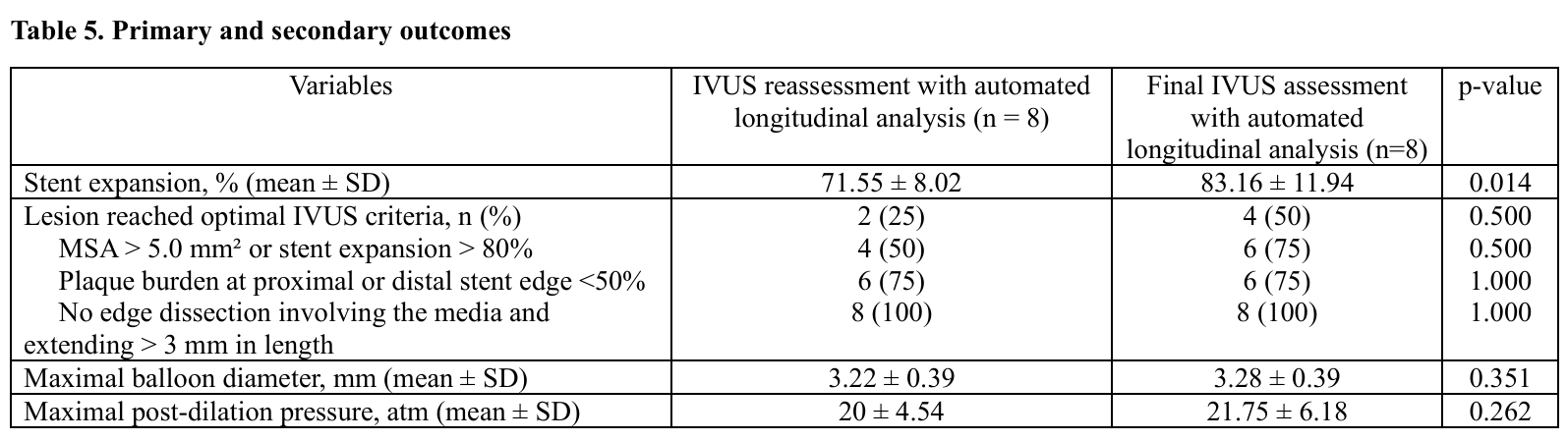
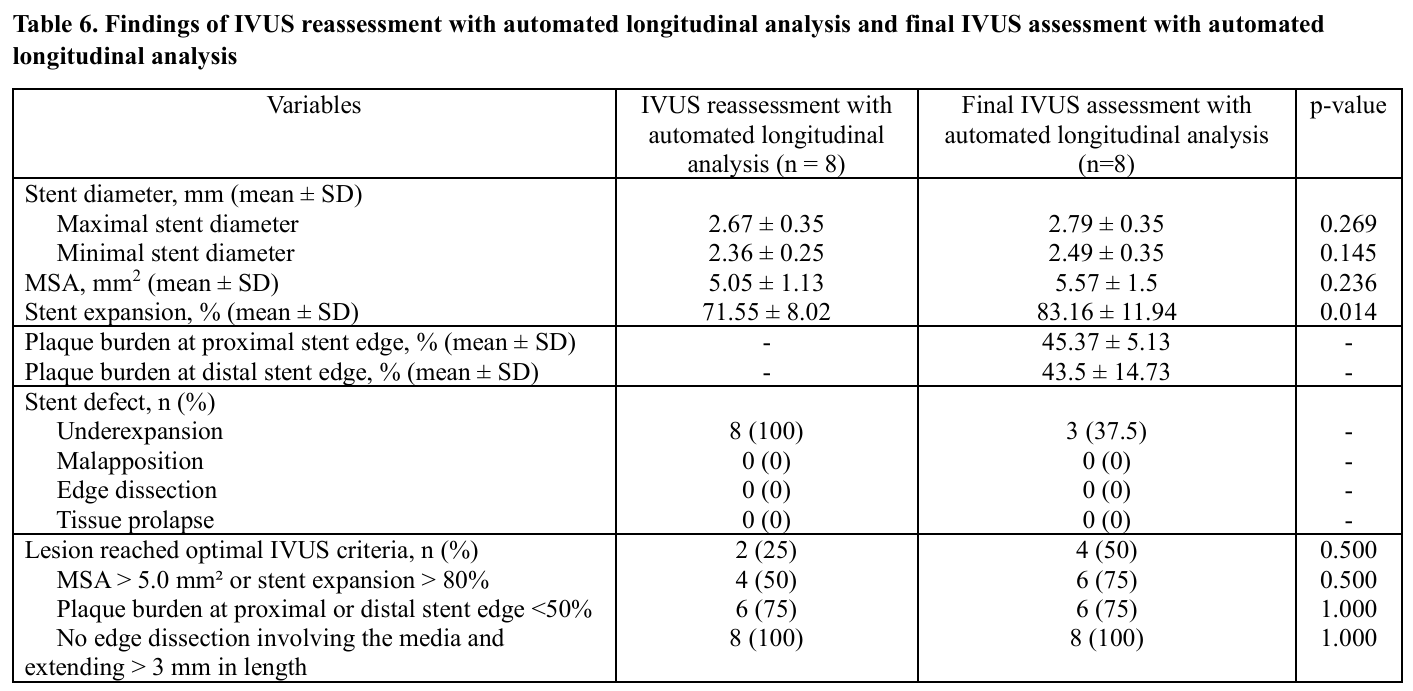
This single-center, single-arm, pre–post interventional study was conducted at Police General Hospital, Bangkok, Thailand. We prospectively enrolled adult patients (≥18 years) who underwent IVUS-guided PCI between September 2024 and March 2025 for chronic coronary syndrome (CCS), non-ST elevation myocardial infarction (NSTEMI) or unstable angina. Exclusion criteria included hemodynamic instability, sustained ventricular arrhythmia, chronic total occlusion, heavily calcified lesions requiring atherectomy, complex bifurcation requiring two-stent technique and prior PCI or CABG involving the target vessel. All procedures followed standard clinical practice. Drug-eluting stents were implanted in all cases. After initial stent deployment and post-dilation, IVUS imaging was performed using a 60 -MHz TrueVision IVUS system (Insight Lifetech, Shenzhen, China) with automated pullback at 1 – 3 mm/s. Operators performed conventional IVUS analysis using manual border tracing. Optimal deployment was defined as: (1) MSA > 5.0 mm² or expansion > 80%; (2) plaque burden < 50% within 5 mm of stent edges; and (3) no edge dissection > 3 mm involving media. Underexpanded stents underwent additional post-dilation followed by repeat IVUS until optimal expansion was achieved.The same IVUS dataset was then reanalyzed using automated longitudinal analysis software, which delineated vessel contours and generated continuous profiles of lumen and stent areas. The system flagged suboptimal regions with low lumen area or high plaque burden. If inadequate expansion was identified, additional post-dilation was performed, followed by repeat automated IVUS assessment.The primary outcome was stent expansion improvement between the automated reassessment and final automated IVUS analysis. Secondary outcomes included the proportion of lesions fulfilling all optimal IVUS criteria and comparisons of balloon size and inflation pressure before and after additional post-dilation.



Results
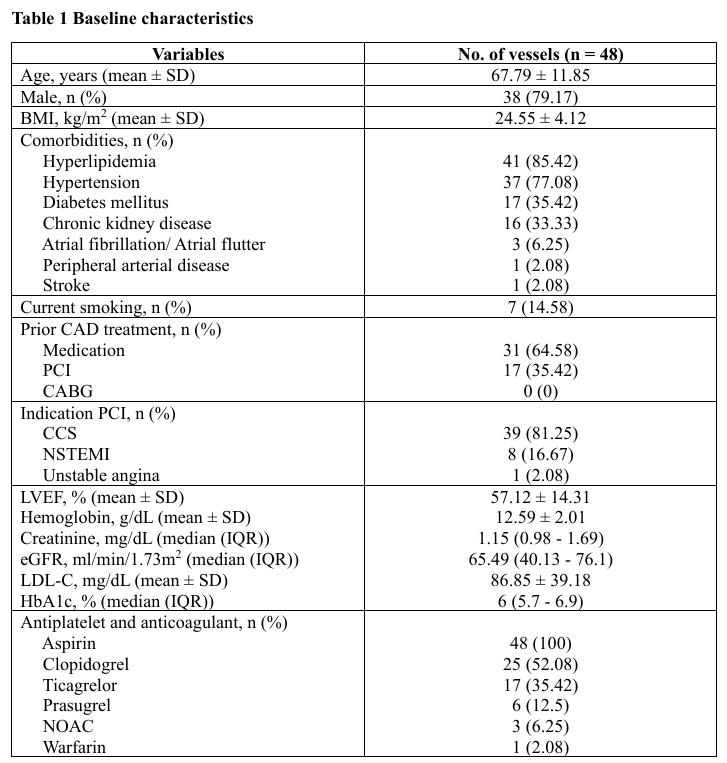
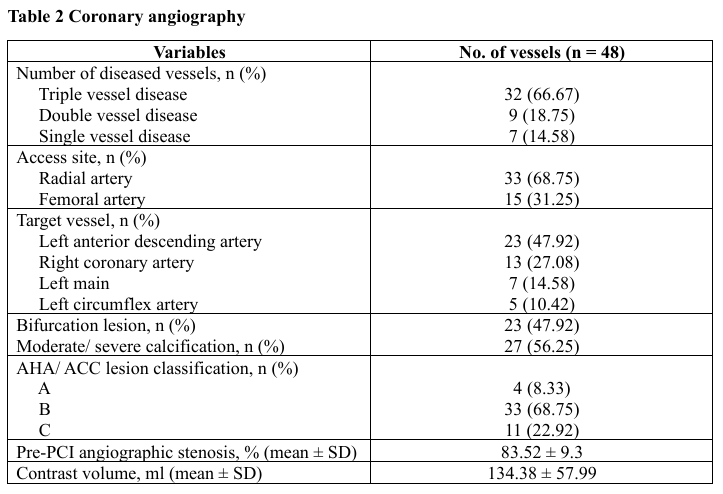
A total of 48 vessels were evaluated. The mean age was 67.8 ± 11.9 years and 79.2% were male. Mean BMI was 24.6 ± 4.1 kg/m². Comorbidities included hyperlipidemia (85.4%), hypertension (77.1%), diabetes mellitus (35.4%) and chronic kidney disease. (33.3%) 14.6% were current smokers. A prior PCI history was present in 35.4%. Most patients underwent PCI for chronic coronary syndrome (81.3%) followed by NSTEMI (16.7%) and unstable angina (2.1%). The mean LVEF was 57.1 ± 14.3%.Angiographically, triple-vessel disease occurred in 66.7%, while single- and double-vessel disease were seen in 14.6% and 18.8%, respectively. The radial approach was used in 68.8%. Target vessels included the LAD (47.9%), RCA (27.1%), LM (14.6%) and LCx (10.4%). Bifurcation lesions were found in 47.9% and moderate-to-severe calcification in 56.3%. According to AHA/ACC classification, type B lesions predominated (68.8%) followed by type C (22.9%) and type A (8.3%). Mean pre-PCI stenosis was 83.5 ± 9.3%.Pre-PCI IVUS revealed mixed plaque (80.9%) and calcified plaque (17.0%). Mean minimal lumen area (MLA) was 2.53 ± 0.92 mm² with lesion length 37.8 ± 17.4 mm. Proximal reference lumen area averaged 9.33 ± 5.05 mm² (plaque burden 47.5 ± 9.8%) and distal reference lumen area 6.09 ± 3.00 mm² (plaque burden 37.7 ± 12.9%). The mean stent number per lesion was 1.52 ± 0.62 with total stent length 40.4 ± 17.7 mm.Compared with conventional IVUS analysis, automated longitudinal analysis yielded smaller measurements of maximal stent diameter (2.79 ± 0.48 mm vs 2.95 ± 0.59 mm, p < 0.001), minimal stent diameter (2.40 ± 0.40 mm vs 2.54 ± 0.50 mm, p < 0.001), MSA (5.43 ± 1.95 mm² vs 6.04 ± 2.58 mm², p < 0.001) and stent expansion (85.5 ± 14.2% vs 95.3 ± 17.3%, p < 0.001). Underexpansion was more frequently detected by automated analysis (27.1% vs 14.6%, p = 0.031).Among eight vessels requiring additional post-dilation, mean stent expansion improved significantly from 71.6 ± 8.0% to 83.2 ± 11.9% (p = 0.014). Lesions meeting all optimal IVUS criteria increased from 25% to 50%, without statistical significance. Balloon diameter and inflation pressure showed non-significant increases.


Conclusion
This study demonstrated the clinical utility of automated longitudinal IVUS analysis in enhancing stent optimization following conventional IVUS-guided PCI. Although conventional IVUS interpretation by experienced operators resulted in better procedural results, automated analysis revealed higher rate of underexpansion, suggesting that subtle areas of suboptimal deployment may be underrecognized by conventional IVUS alone.IVUS with automated longitudinal analysis enhances the precision of stent assessment and facilitates identification of suboptimal deployment following PCI. In this study, additional optimization guided by automated analysis significantly improved stent expansion, suggesting its potential as a valuable adjunct to conventional IVUS. Despite being underpowered, our findings support the role of this technology in standardizing intravascular imaging and improving the consistency and quality of PCI outcomes.
