Lots of interesting abstracts and cases were submitted for TCTAP 2026. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge!
ABS20250929_0002
Modeling Heart-Brain Crosstalk Under SGLT2 Inhibition: A Digital Twin Approach Using Transcriptomic and Ligand-Receptor Simulation
By Javad Alizargar
Presenter
Javad Alizargar
Authors
Javad Alizargar1
Affiliation
National Taipei University of Nursing and Health Science, Taiwan1
View Study Report
ABS20250929_0002
Digital Health and Artificial Intelligence
Modeling Heart-Brain Crosstalk Under SGLT2 Inhibition: A Digital Twin Approach Using Transcriptomic and Ligand-Receptor Simulation
Javad Alizargar1
National Taipei University of Nursing and Health Science, Taiwan1
Background
The heart–brain axis plays a crucial rolein cardiovascular and neurological health, yet the mechanisms underlying itsregulation by pharmacologic agents remain poorly defined. Sodium–glucosecotransporter 2 inhibitors (SGLT2i), originally developed for glycemic control,have demonstrated unexpected benefits across heart, kidney, and brain systems,raising interest in their inter-organ effects. We aimed to simulate andmechanistically model heart–brain communication under SGLT2 inhibition usingtranscriptomic data and a digital twin framework.
Methods
Using publicly available transcriptomicdatasets from empagliflozin-treated murine cardiac (GSE270093) and rat braintissue (GSE206986), we performed differential gene expression and pathwayenrichment analyses. Ligand–receptor interactions between heart-derived ligandsand brain-expressed receptors were mapped and simulated under knockout andoverexpression conditions. Receptor-weighted pathway propagation models andbrain single-cell transcriptomic data (DropViz) were integrated to inferdownstream signaling effects. A network model was constructed to visualize coreinter-organ signaling routes.
Results
Empagliflozin downregulated cardiac stressgenes (e.g., Nppa, Nppb), fibrotic markers (Col1a1, Timp1), and inflammatorycytokines (Il1b, Cxcl10), while upregulating sympathetic and mitochondrialgenes. Brain transcriptomes revealed upregulation of NF-κB and toll-likereceptor pathways. Cross-tissue analysis identified key ligand–receptor axes(e.g., CXCL2→CXCR2, BDNF→INSRR) that linked cardiac output to brain receptoractivation. Simulated perturbations revealed fragile vs. amplifiable signalingroutes. DropViz analysis confirmed cell-type–specific receptor localization inneurons and glia. A final network diagram highlighted integrated cytokine,neurotrophic, and metabolic communication across the heart–brain axis.
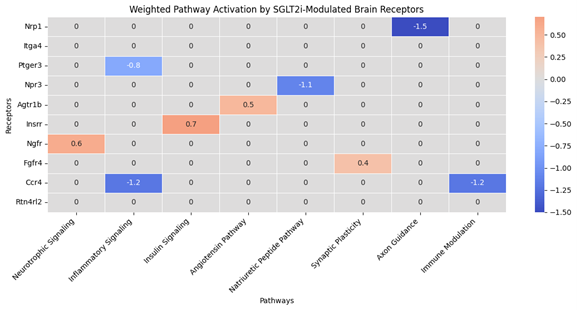
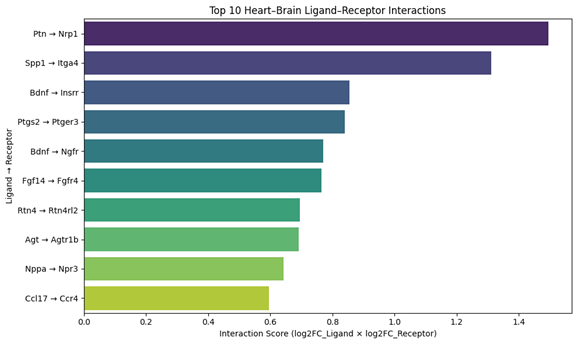
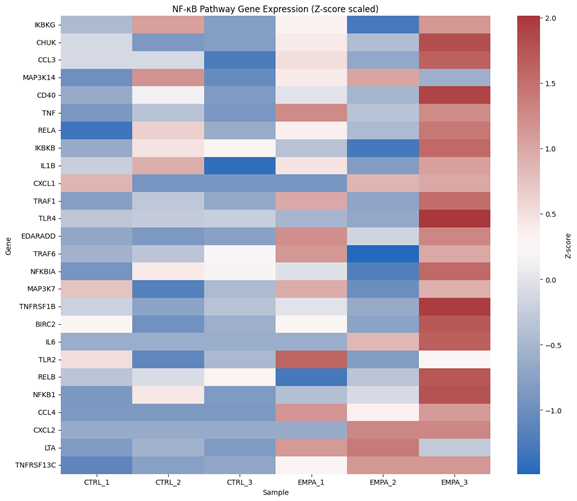



Conclusion
SGLT2 inhibition alters heart-derivedmolecular signals that modulate brain pathways through defined ligand–receptorinteractions. This transcriptomics-informed digital twin model revealsstructured, cell-type–specific, and targetable inter-organ signaling networks,offering a scalable platform to explore cardiometabolic–neuroprotectivetherapies.
