An expert outlined key takeaways on using fractional flow reserve (FFR) and pressure pullback gradient (PPG) after percutaneous coronary intervention (PCI) to predict post-revascularization clinical outcomes.
 “Post-PCI FFR is nothing new, and was used to assess coronary artery stenosis and post-PCI physiology since the late 1970s” Carlos Collet, MD, PhD(OLV Hospital, Aalst, Belgium) said at TCTAP 2022 on Apr 29. “The question is whether post-PCI FFR can be a marker of prognosis or is just a bystander in the relationship between coronary artery disease (CAD) pattern and major adverse cardiovascular events (MACE).”
“Post-PCI FFR is nothing new, and was used to assess coronary artery stenosis and post-PCI physiology since the late 1970s” Carlos Collet, MD, PhD(OLV Hospital, Aalst, Belgium) said at TCTAP 2022 on Apr 29. “The question is whether post-PCI FFR can be a marker of prognosis or is just a bystander in the relationship between coronary artery disease (CAD) pattern and major adverse cardiovascular events (MACE).”
FFR is an imaging tool used to assess lesion significance and characterize disease before stent implantation to optimize PCI. PPG is a novel metric that helps identifies CAD phenotypes on a scale of 0 (diffuse disease) to 1 (focal disease) based on FFR pullbacks and tailor revascularization strategies.
Link between low post-PCI FFR and MACE: causality or association?
Several studies have identified a relationship between post-PCI FFR and factors like clinical outcomes, clinical characteristics, and CAD patterns.
An individual patient-level meta-analysis on 2,400 patients by Collet and investigators, currently under review, found that lower post-PCI FFR increased the risk of target-vessel failure (TVF, OR per 0.10 FFR units: 0.56, 95% CI, 0.45-0.69, p<0.001) as well as myocardial infarction (MI) and cardiac death (aHR 0.67, 95% CI, 0.46-0.98, P=0.037).
Further studies found post-PCI FFR was improved significantly in focal CAD, but not in diffuse CAD. A study by Mizukami and investigators employing PPG demonstrated that focal disease had significantly improved blood flow than diffuse disease and the disease type determined half of the blood flow improvement achieved with PCI (R2=51%).
Collet’s individual patient-level meta-analysis (under revision) on 2,760 patients and 3,336 post-PCI FRR measurements found post-PCI FFR of the left anterior descending (LAD) artery was significantly lower than that of other vessels (0.06 FFR, 95% CI, 0.05-0.08, p<0.001), indicating predictive capacity of post-PCI FFR differs depending on LAD or non-LAD.
Regarding post-PCI FFR’s predictive value for TVF stratified by LAD or non-LAD, investigators found the best cutoff for non-LAD was 0.90 (AUC 0.61; 95% CI, 0.56-0.67), which was higher than LAD’s 0.80 (AUC 0.54; 95% CI 0.48-0.59). Patients with high post-PCI FFR also had improved prognosis for TVF, irrespective of the vessel treated, when using the 0.80 differential cutoff for TVF in LAD and 0.90 for non-LAD.
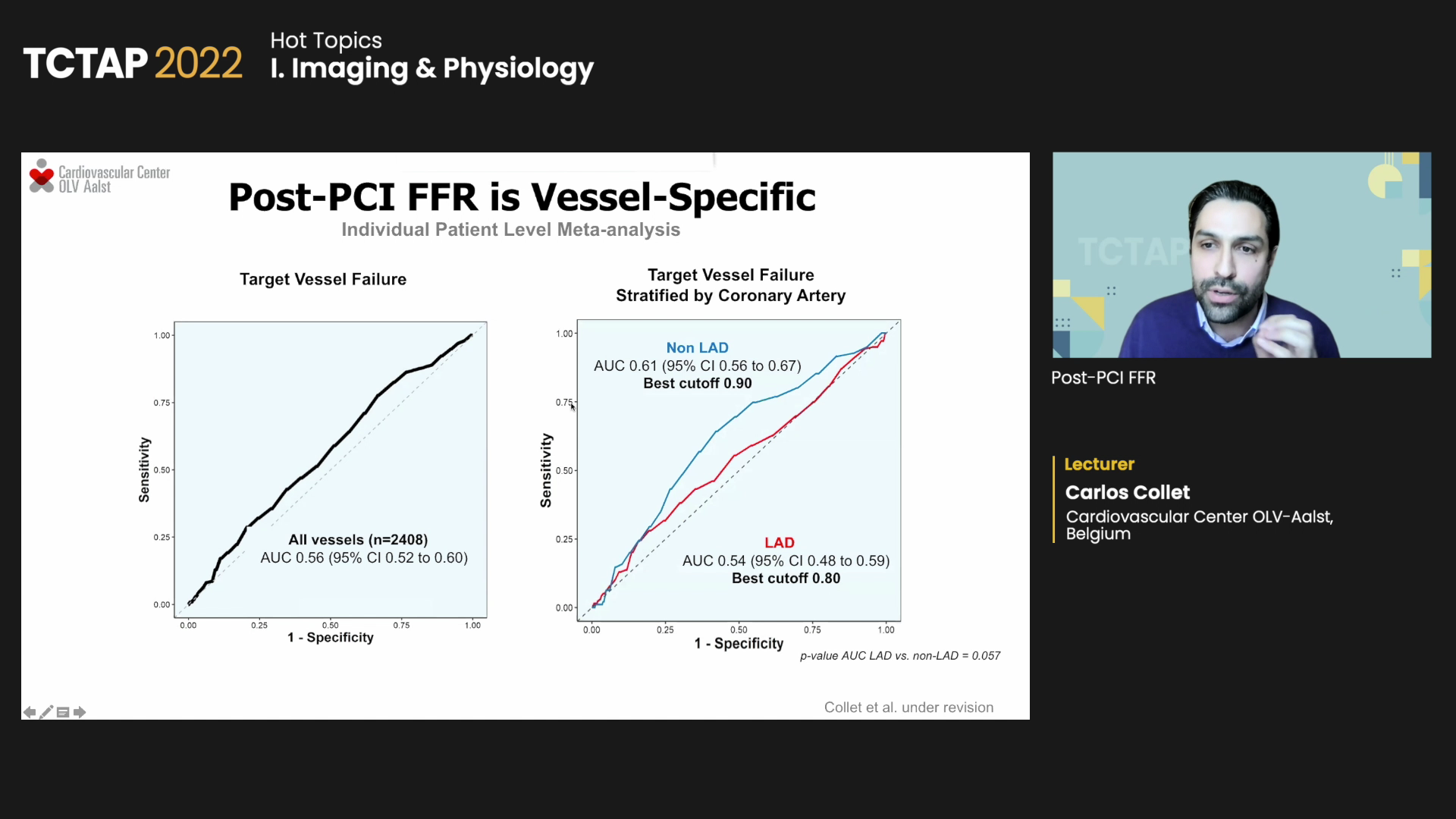
“Post-PCI FFR is an independent predictor of MACE, but with moderate predictive capability for events,” Collet said. “Post-PCI FFR should be interpreted in a vessel-specific manner since studies show post-PCI FFR in LAD was lower (0.06 FFR units) than in non-LAD.”
Low post PCI FFR can be classified as in-stent focal gradient, focal gradient outside the stent, and diffuse pattern, all of which require different treatment, he continued.
In-stent focal pressure gradients are related to stent under-expansion and call for post-dilatation. Focal gradients outside the stent are associated with plaque stress or fatigue that could cause eventual plaque rupture, entailing treatment of vulnerable plaques or additional stent implantation. For diffuse pattern, optimal medical therapy is the best option.
Remaining questions in the post-PCI FFR landscape include whether treating patients with low post-PCI FFR improves clinical outcomes as well as the relationship between post-PCI FFR and MACE, specifically whether diffuse and focal residual pressure gradients have the same clinical significance for adverse clinical events.
“Post-PCI FFR depends on the baseline CAD pattern, and is associated with clinical outcomes,” Collet said. “Post-PCI FFR should be interpreted in a vessel-specific manner, but more research is needed to understand if additional treatment in response to post-PCI FFR translates to better clinical outcomes.”
Edited by

Se Hun Kang, MD
CHA Bundang Medical Center, Korea (Republic of)
Written by



